Beta Globin Protein Structure
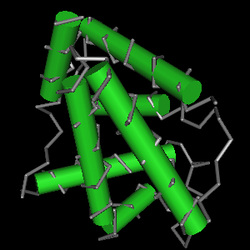
The messenger RNA (mRNA) code was also located in the Nucleotide database and is 626 nucleotides in length (NM_000518.4) (Nucleotide, NCBI- Human HBB mRNA, 2011). After the introns are spliced out of the pre-mRNA, the mature mRNA is translated into the human β-globin polypeptide which is 147 amino acids in length (NP_000509.1) (Protein-NCBI, 2011b). The human β-globin’s secondary structure is in the form of multiple alpha helices attached by non-helical segments (Fig. 1). The helical segments are formed by hydrogen bonds (Hardison, 1996). Normal folding of β-globin into this alpha helix allows for the formation of the tertiary structure of each of the globin molecules.
Figure 1: Beta Globin molecule
The quaternary structure produces the mature hemoglobin molecule containing two α-globin chains and two β-globin chains. An iron containing heme molecule is tightly bound to each polypeptide. One oxygen molecule binds to each heme molecule (Hardison, 1998; US Library of Medicine, 2011) (Fig. 2).
Figure 2: Quaternary Protein Structure of Human Hemoglobin. (MNIMBS, 2010)
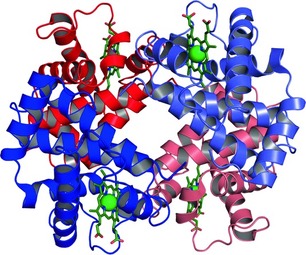
Red Molecules represent α-globin
Blue molecules represent β-globin
Green molecules symbolize heme
Blue molecules represent β-globin
Green molecules symbolize heme
